What's in this lesson
Master the concept of Resonance, structural stability rules, and the SIR effect crucial for JEE Advanced.
Why this matters (WIIFM)
Resonance explains the fundamental stability, acidity, and reactivity of aromatic and conjugated organic molecules. Without it, predicting reaction pathways is impossible.
Attention Activity: The Benzene Mystery
Benzene (C₆H₆) is classically drawn with alternating single and double bonds. Yet, experimental data shows all carbon-carbon bonds are exactly equal!

Observation: All C-C bonds in Benzene are 139 pm long, an intermediate between a single (154 pm) and double (134 pm) bond. This is the magic of Resonance!
What is Resonance?
Resonance is the phenomenon where π (pi) electrons are delocalized across a network of adjacent, parallel p-orbitals.

Click to Compare: Localized vs Delocalized
Localized: Electrons confined between two specific atoms.
Delocalized: Electrons shared across 3 or more atoms, lowering overall energy.
Excellent! Delocalization fundamentally stabilizes the molecule.
Conditions for Resonance: Conjugation
For electrons to delocalize, the molecule must have a conjugated system—a continuous chain of sp² or sp hybridized atoms.
Drag the "π electrons" block into the conjugated system box to initiate resonance.
Conjugation achieved! Common types include π-π, π-positive, and π-lone pair conjugations.
Rules for Drawing Resonating Structures
Resonance strictly involves the movement of electrons, never atoms. Follow these sequential rules.
Never move atoms, only π electrons or non-bonding lone pairs.
Total formal charge of the molecule must remain constant.
Number of unpaired electrons must remain the same.
Knowledge Check
Which of the following is a strict requirement for resonance to occur?
Stability Rule 1: The Octet Rule
Not all resonance structures contribute equally. The most dominant structure has the highest stability.
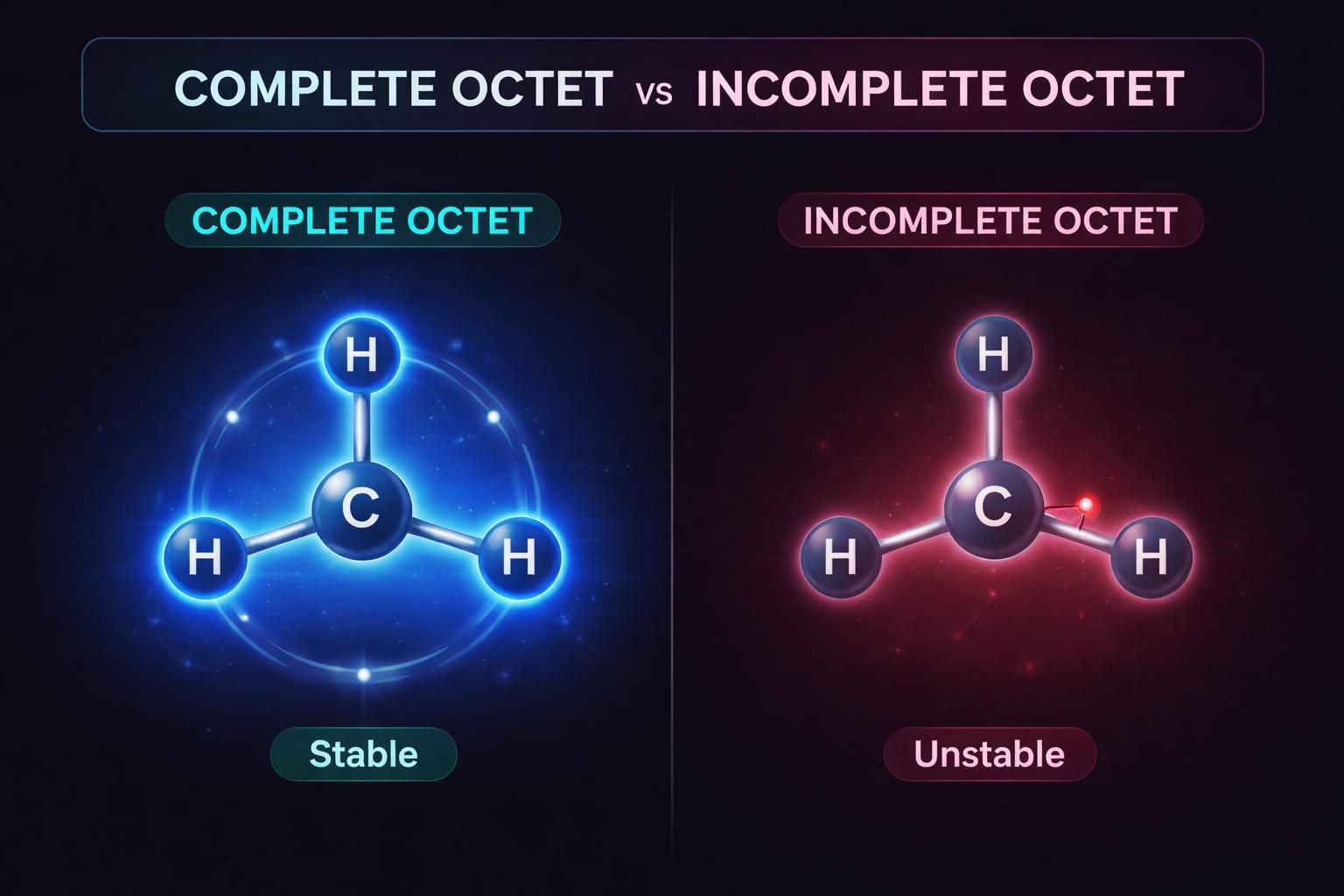
Structures with a greater number of covalent bonds and complete octets for every atom are inherently more stable.
Even if an atom carries a formal positive charge (like Oxygen in an oxonium ion), if its octet is complete, the structure is highly stable and often the major contributor.
Stability Rule 2: Charge Distribution
Charge separation requires energy. Therefore, neutral structures are almost always more stable than those with formal charges.

If formal charges must exist, structures where unlike charges (+ and -) are close together are more stable due to electrostatic attraction. Like charges should be as far apart as possible.
Stability Rule 3: Electronegativity
When negative charges must exist, they are most stable on highly electronegative atoms (like Oxygen or Nitrogen).
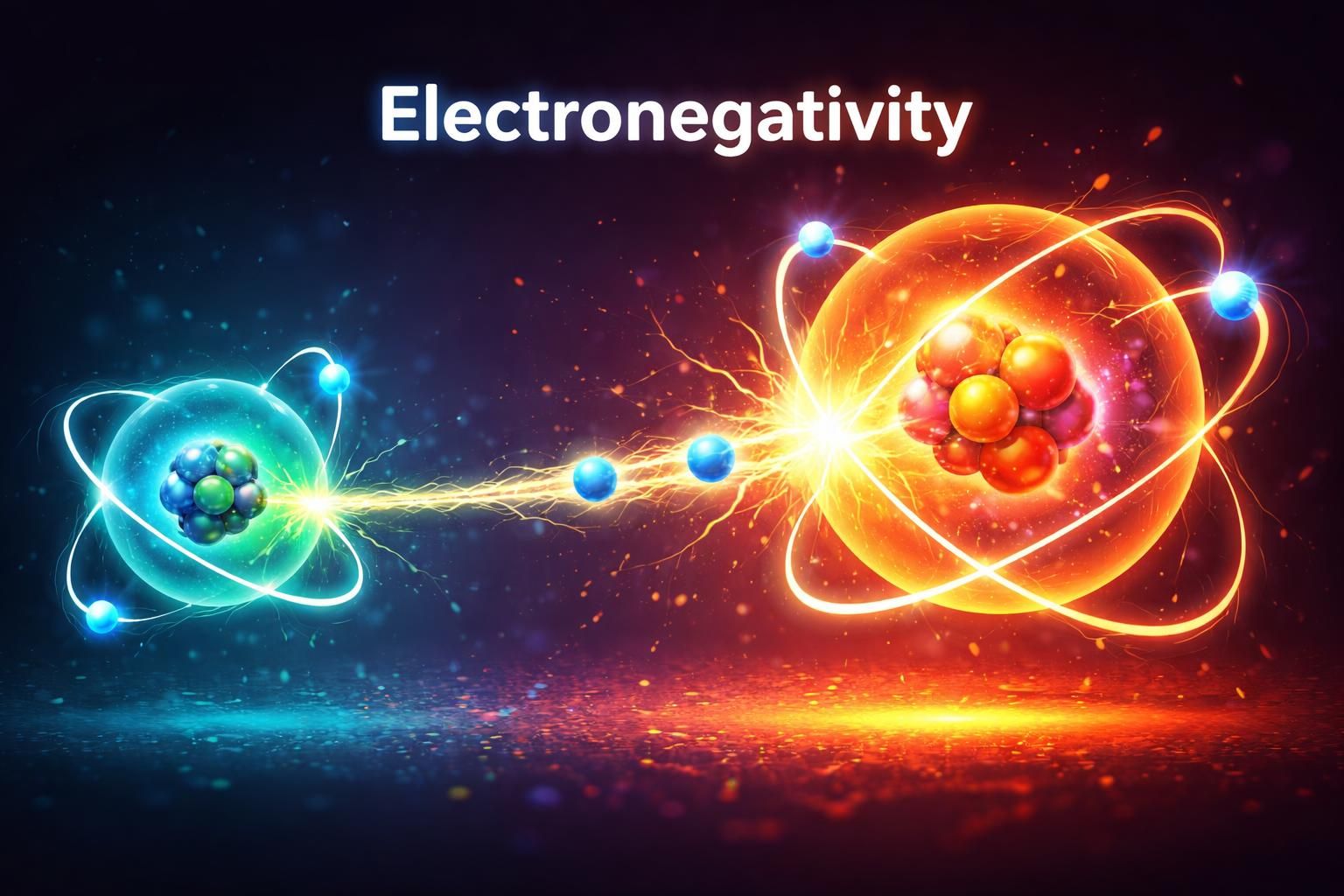
Correct! The enolate resonance structure with the negative charge on Oxygen is the major contributor compared to the charge on Carbon.
Knowledge Check
Among the resonance structures of an enolate ion, which factor primarily dictates the major contributor?
The Resonance Hybrid
Individual resonance structures don't actually exist in reality. The true molecule is a Resonance HybridA weighted average of all valid structures.

The hybrid is always more stable than any single contributing structure. It perfectly explains observed physical properties.
Resonance Energy
The extra stability gained by delocalization is called Resonance Energy (RE). It is the energy difference between the actual hybrid and the most stable theoretical structure.
Benzene's Resonance Energy is approx. 152 kJ/mol, making it exceptionally stable!
Applications: Bond Length Variations
Resonance gives single bonds partial double bond character (shortening them) and gives double bonds partial single bond character (lengthening them).
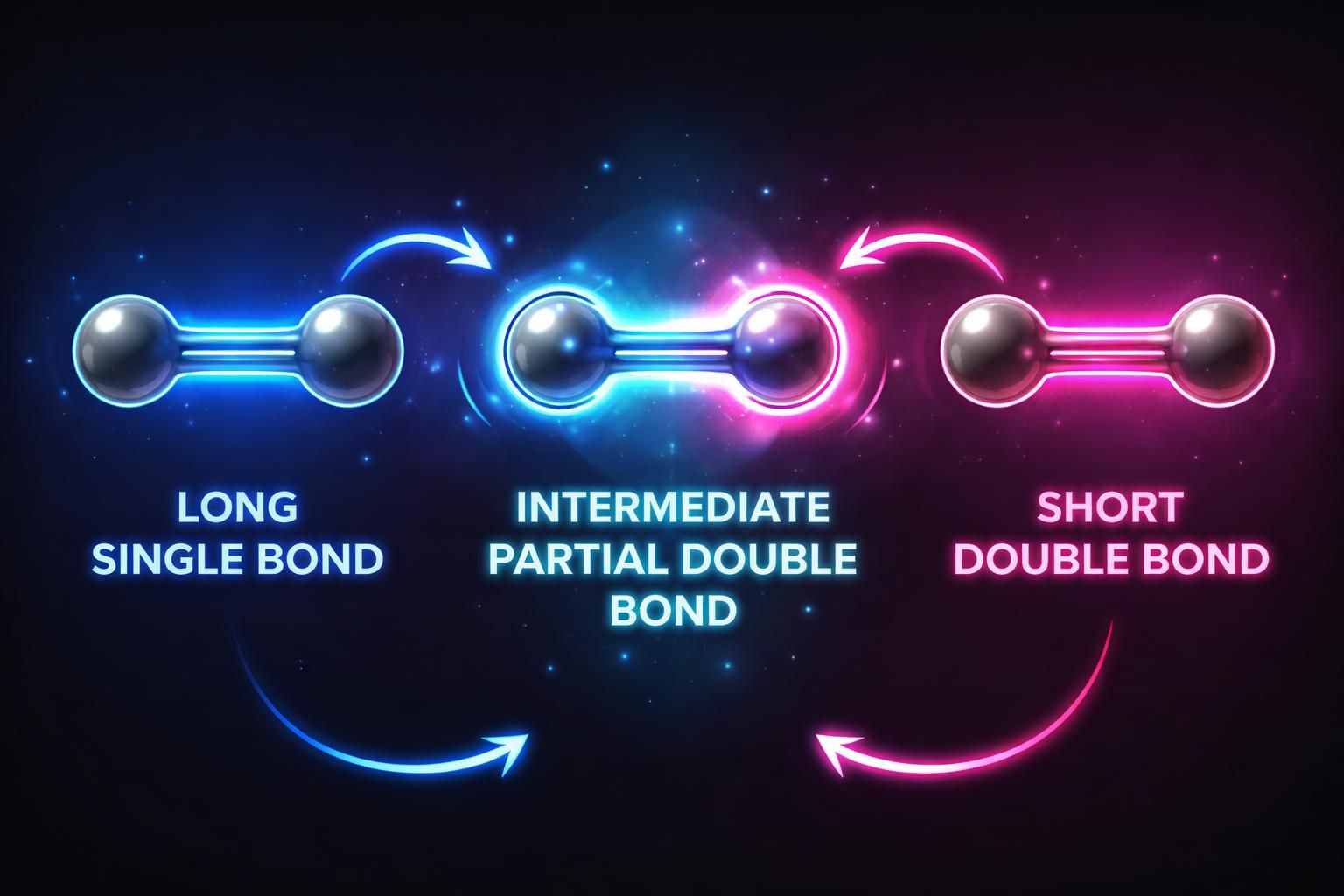
139 pm: The perfect intermediate length found in Benzene due to resonance!
Steric Inhibition of Resonance (SIR Effect)
In JEE, you must watch out for the SIR effect. Bulky substituent groups can physically twist the conjugated system out of plane, destroying parallel p-orbital overlap.

Knowledge Check
How does the SIR effect influence resonance in a molecule?
Key Takeaways
- Conjugation is King: Resonance strictly requires parallel, overlapping p-orbitals.
- Stability Hierarchy: Complete octets > Minimal charge separation > Negative charge on electronegative atoms.
- The Hybrid Reality: The true molecule is a weighted resonance hybrid, uniquely stabilized by Resonance Energy.
- The SIR Effect: Steric crowding can break planarity, instantly terminating resonance and radically altering chemical properties.
Final Assessment
You are about to begin the final JEE-style assessment on Resonance.
There are 5 questions. You need 80% to earn your completion certificate.
Good luck!
Assessment Question 1
Which condition is essential for resonance to occur in a molecule?
Assessment Question 2
When evaluating the stability of resonating structures, which rule generally takes highest priority?
Assessment Question 3
The actual structure of a molecule experiencing resonance is best described as:
Assessment Question 4
What does Resonance Energy represent in the context of organic chemistry?
Assessment Question 5
What is the primary consequence of Steric Inhibition of Resonance (SIR)?
Assessment Submitted
Calculating your JEE readiness score...